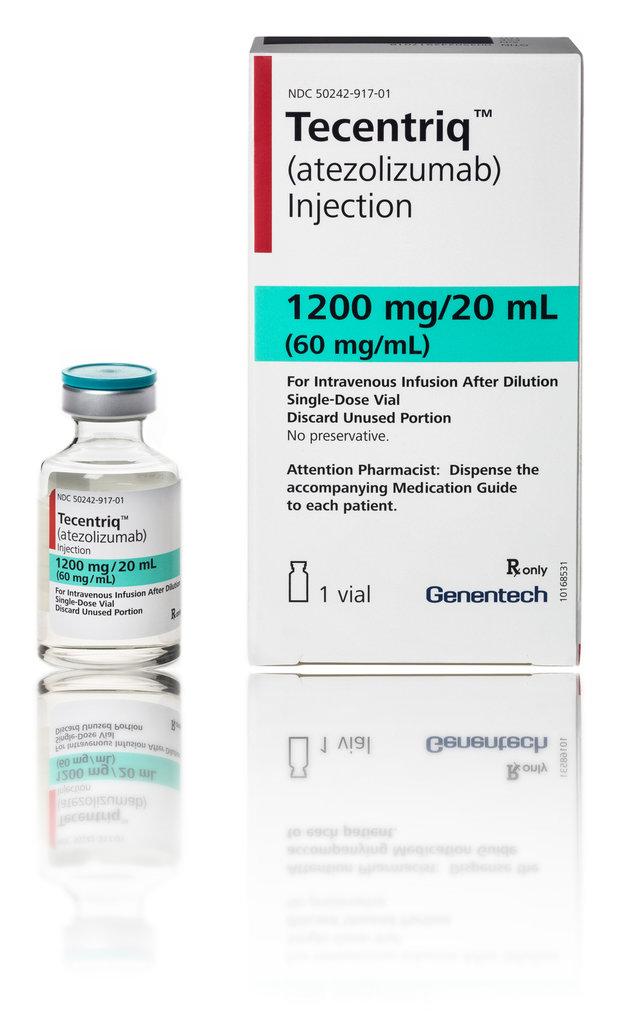
Lung and Bladder Cancer drug is launched by Roche Products India Pvt Ltd, a major Global pharma company. Today, It said that it has launched monoclonal antibody atezolizumab under the brand name Tecentriq in India for treatment of lung and bladder cancer. “Tecentriq has been proved to extend the time of life and thus improving the quality of life. This product is approved in India for non-small cell lung cancer (NSCLC) and Urothelial Carcinoma, a type of bladder and urinary tract cancer", said Roche.The drug, called Tecentriq, is the fourth medicine of a new class called checkpoint inhibitors that work by unleashing the body’s immune system to attack tumors. Adding Roche's immunotherapy Tecentriq to older drugs doubled the percentage of lung cancer patients who survived a year without their disease advancing, an outcome some experts labeled unprecedented.Thirty-seven percent of patients in a closely watched clinical trial who got Tecentriq, Avastin and chemotherapy reached the one-year mark without their cancer progressing (PFS).Patients getting Roche's immunotherapy survived an average of 8.3 months without their disease getting worse, compared with a PFS of 6.8 months for those getting chemotherapy and Avastin. On the prescription of Oncologists, this drug will be availed to patients. It is believed that Tecentriq is set for the major goals in the treatment of advanced bladder cancer and metastatic non-small cell lung cancer in India, she added. "Roche has helped transform the treatment of cancer globally, and will continue to set newer standards of care for cancer patients in India," Bezerra said. Managing Director Lara Bezerra said, “Bringing Tecentriq to India reflects Roches commitment to bring global innovations to the country." Tecentriq is a monoclonal antibody used for treatment of metastatic non-small cell lung cancer and metastatic urothelial carcinoma. It is a cancer immunotherapy drug that is designed to work with body's natural defense system. She said, “Right diagnosis is very important for immunotherapy, and we are looking at how we can use technology in the right direction to identify more patients.”
Roche launches new lung, bladder cancer drug in India